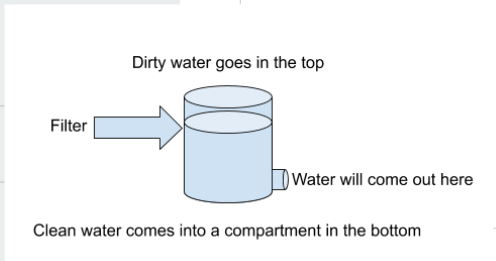
Our Task:
We started by asking some guiding questions to help us understand where we wanted to go with our project.
Example questions that we considered:
Example questions that we considered:
|
What kind of filters are we going to use?
What kind of chemicals will we use? What would the price look like for this item? How will it be accessible to people in developing countries? How will it be sustainable or environmentally friendly? |
What kind of table display will convey our message or problem?
How large should the item be? How much will it weigh? What things that are similar to this currently exist? |
We then looked into the problem we hoped to address through working on this project. We decided that we hoped to help third world countries successfully gain access to clean drinking water that is affordable. With our model we hope to provide people without clean water with an easy solution so they can stay healthy and live prosperous lives.
Why this issue?
|
Background:
Before we jumped into this project, we did some background research to learn more about how dirty water works. We began by learning about the Flint water crisis. In the process of building a new pipeline to bring water from Lake Huron to the city of Flint, Michigan, water became foul smelling and discolored. We learned that the protective membrane on the pipes in Flint had eroded away and that lead from the pipes had begun to get into the water. This water also had high levels of organic molecules called trihalomethanes which can get into the water when chlorine is used to disinfect. This chlorine reacts with algae and leaves to produce trihalomethanes. These can lead to future health problems such as liver, kidney, and heart conditions.
Our Project:
We began with the idea of a filter. In a filter, water passes through screens made of mesh or other polymers to remove large particles such as sticks, leaves, and other debris. In the next stage of a filter, the water passes through a series of filters consisting of sand and sometimes even biological filters such as algae to remove smaller contaminants, such as dangerous bacteria. Finally, filters can have a disinfection stage where chemicals such as chlorine can be used to treat the water.
Our final filter prototype has a gravel layer for filtering out large particles, a sand layer for finer particles, a activated charcoal layer, and a cotton ball filter layer to keep all of the materials in place.
Our Presentation:
Content:
Solution: A solution is a mixture composed of two or more substances including a solvent and a solute. In our project, the dirty water sample we create was a solution consisting off mud, water, and various other materials.
Solute: A solute is a component of a solution that gets dissolved into the solvent. For example, in a salt water solution, the salt would be the solute that would be dissolved into the water, the solvent.
Solvent: A solvent is a component of a solution that has the ability to dissolve other substances. As discussed in the example above, water could be the solvent in a salt water solution.
Ionic: An ionic bond is created by oppositely charged ions. Ionic bonds involve anions and cations which are negatively and positively charged ions.
Molecular: A molecular bond is a bond between two atoms that share electrons. The sharing of electrons can help the different atoms to fill their outer shells to complete the outer octet of electrons.
Polarity: Polarity is the separation of electrical charges so an object will have a positive charged end and a negative charged end. The negative charge or polarity of charcoal helps it attract toxins, which are positively charged and of the opposite polarity.
Solubility: Solubility is how well the solute is able to dissolve in the solvent.
Organic mater in water bacteria & virus: Besides dirt and other substances that can get into the water, bacteria and viruses can get into water sources. These are much harder to filter out and require chemical reactions to remove. In our project, we used the chemical chloramine to remove bacteria and viruses from the water. These substances can cause many health issues when not removing including death making them a high priority when treating water.
Properties of Acids & Bases: Bases are usually much soapier in nature and have a bitter taste to them. Bases have a pH greater than 7 while acids will have a pH lower than 7. Acids are much more sour and have greater corroding abilities. When an acid reacts with a base, a salt is formed. Acids and based can also both conduct electricity.
pH & what it measures, pH scale, drink-ability range: pH is the potential of hydrogen and the pH scale is a scale of pH levels. This scale ranks how basic or acidic substances are. It is based on the amount of hydrogen ion activity within a certain substance. A high pH means there is a low concentration so it is basic and a low pH means there is a high concentration so it is acidic.
What tests did you conduct:
Ammonia Nitrogen Test: The Ammonia Nitrogen levels stayed the same for the dirty sample before and after filtering at around 0.1 ppm which is within the safe levels. Ammonia is dangerous to aquatic life but it is fine in human drinking water.
pH Levels: The dirty sample had a pH of 7 and the filtered had a pH of 8 while the safe pH levels for drinking water are between 6- 8.5.
Iron Test: The dirty sample had 10 ppm while the filtered sample had 5 ppm for iron. Safe levels of iron in drinking water should be less than 0.3 ppm so we would like to further look into lowering iron levels. Iron in drinking water contains bacteria that feeds off of it and high concentrations of iron can lead to overload which causes diabetes, stomach problems, vomiting and nausea.Copper Test: There was no copper present in either of our samples.
Chlorine Test: There was no chlorine present in either of our samples.
Activated charcoal: Activated charcoal is created by changing the internal structure of charcoal through high temperatures. This change in structure comes in the form of smaller pores. Activated charcoal has a negative electrical charge which helps it attract positively charged molecules such as toxins that can be found in water. Through this attraction, these toxins are absorbed by the charcoal. It is still safe to consume this charcoal because it is not absorbed by our bodies. This means that it will carry the absorbed toxins in and out of our digestive tract without any harm. Activated charcoal is not good at removing bacteria or viruses, but, many organic impurities in water are carbon based and attracted to the carbon in the charcoal creating chemical bonds between the two. Other compounds, such as water, do not bond to the charcoal so they are able to pass through.
Chloramine: Chloramine is created by bonding ammonia with chlorine. It is good at killing off germs and disinfecting drinking water. In waters of pH 7 or higher, monochloramines form which are used in drinking water and are safe for humans to consume. The idea pH for chloramine to form is 8 so this chemical would work fine in our filter because our water samples had a pH of 8 as well. This chemical is more beneficial than chlorine because it lasts much longer than chlorine. Reaction equation: NH3 + HOCI -> NH2Cl + H2O
Solute: A solute is a component of a solution that gets dissolved into the solvent. For example, in a salt water solution, the salt would be the solute that would be dissolved into the water, the solvent.
Solvent: A solvent is a component of a solution that has the ability to dissolve other substances. As discussed in the example above, water could be the solvent in a salt water solution.
Ionic: An ionic bond is created by oppositely charged ions. Ionic bonds involve anions and cations which are negatively and positively charged ions.
Molecular: A molecular bond is a bond between two atoms that share electrons. The sharing of electrons can help the different atoms to fill their outer shells to complete the outer octet of electrons.
Polarity: Polarity is the separation of electrical charges so an object will have a positive charged end and a negative charged end. The negative charge or polarity of charcoal helps it attract toxins, which are positively charged and of the opposite polarity.
Solubility: Solubility is how well the solute is able to dissolve in the solvent.
Organic mater in water bacteria & virus: Besides dirt and other substances that can get into the water, bacteria and viruses can get into water sources. These are much harder to filter out and require chemical reactions to remove. In our project, we used the chemical chloramine to remove bacteria and viruses from the water. These substances can cause many health issues when not removing including death making them a high priority when treating water.
Properties of Acids & Bases: Bases are usually much soapier in nature and have a bitter taste to them. Bases have a pH greater than 7 while acids will have a pH lower than 7. Acids are much more sour and have greater corroding abilities. When an acid reacts with a base, a salt is formed. Acids and based can also both conduct electricity.
pH & what it measures, pH scale, drink-ability range: pH is the potential of hydrogen and the pH scale is a scale of pH levels. This scale ranks how basic or acidic substances are. It is based on the amount of hydrogen ion activity within a certain substance. A high pH means there is a low concentration so it is basic and a low pH means there is a high concentration so it is acidic.
What tests did you conduct:
Ammonia Nitrogen Test: The Ammonia Nitrogen levels stayed the same for the dirty sample before and after filtering at around 0.1 ppm which is within the safe levels. Ammonia is dangerous to aquatic life but it is fine in human drinking water.
pH Levels: The dirty sample had a pH of 7 and the filtered had a pH of 8 while the safe pH levels for drinking water are between 6- 8.5.
Iron Test: The dirty sample had 10 ppm while the filtered sample had 5 ppm for iron. Safe levels of iron in drinking water should be less than 0.3 ppm so we would like to further look into lowering iron levels. Iron in drinking water contains bacteria that feeds off of it and high concentrations of iron can lead to overload which causes diabetes, stomach problems, vomiting and nausea.Copper Test: There was no copper present in either of our samples.
Chlorine Test: There was no chlorine present in either of our samples.
Activated charcoal: Activated charcoal is created by changing the internal structure of charcoal through high temperatures. This change in structure comes in the form of smaller pores. Activated charcoal has a negative electrical charge which helps it attract positively charged molecules such as toxins that can be found in water. Through this attraction, these toxins are absorbed by the charcoal. It is still safe to consume this charcoal because it is not absorbed by our bodies. This means that it will carry the absorbed toxins in and out of our digestive tract without any harm. Activated charcoal is not good at removing bacteria or viruses, but, many organic impurities in water are carbon based and attracted to the carbon in the charcoal creating chemical bonds between the two. Other compounds, such as water, do not bond to the charcoal so they are able to pass through.
Chloramine: Chloramine is created by bonding ammonia with chlorine. It is good at killing off germs and disinfecting drinking water. In waters of pH 7 or higher, monochloramines form which are used in drinking water and are safe for humans to consume. The idea pH for chloramine to form is 8 so this chemical would work fine in our filter because our water samples had a pH of 8 as well. This chemical is more beneficial than chlorine because it lasts much longer than chlorine. Reaction equation: NH3 + HOCI -> NH2Cl + H2O
Reflection:
What went well over this project was our ability to create our slide show and presentation. My group was able to quickly create our presentation with efficiency and accuracy so that we were ready to present on the day of presentations. We were all able to collaborate and communicate in order to make sure that our work was done on time. We divided up the work and collaborated on the slides to ensure that we all worked together to produce a final product. We also had to use good communication skills to work on the project outside of class. Our group got assigned to different tutorial classes the day of presentations so we had to use our communication skills to coordinate the final touches that needed to be added and how we planned on presenting our slide show. One aspect that went poorly was trying to find a sample of water to test. We wanted to find a water sample that would be very dirty so it took us awhile to find what we were looking for. We wasted some time doing tests on water samples that we ended up not even using.
I learned that being culturally competent can apply to science. I would have not considered this before but after the water project I have become more aware about different cultures and life styles in different parts of the world. For example I learned much more about the impacts of unclean water and how many cultures throughout the world suffer with this issue on a daily basis. I also learned that it is very hard to find something in the world that has not been done already. Throughout this project we found that many of the ideas we wished to pursue had already been researched and done by many other people. I learned that in order to make something new you have to be a critical thinker and put your own unique spin on something. Our group decided to make a common filter but we included the use of chloramines which is a less traditional purification method making our filter unique.
I will continue to work on spreading my work out evenly so I am not doing all of my work the day before an assignment is due. It is always best to work in smaller intervals instead of cramming in information so on the next project I will work on doing an even workload throughout the week. I can also improve on making sure we allow for more time to organize and practice our presentation. We can always improve on making sure our slide show presentation can flow smoother and this can be accomplished by setting aside one day to focus on practice presentations. A peak from this project was when our filter actually lowered the amount of iron present in the dirty water. All of our other tests did not give us any idea of whether or not our filter was doing anything but the iron levels did actually lower after being filtered. A pit from this project was when our filter broke. The paper that we used to line the bottom of the filter had deteriorated after many uses and much of the dirty water was able to pass through the filter. Even with this setback we were able to continue on and reconstruct a new filter to finish up the last few testes we needed to complete.
I learned that being culturally competent can apply to science. I would have not considered this before but after the water project I have become more aware about different cultures and life styles in different parts of the world. For example I learned much more about the impacts of unclean water and how many cultures throughout the world suffer with this issue on a daily basis. I also learned that it is very hard to find something in the world that has not been done already. Throughout this project we found that many of the ideas we wished to pursue had already been researched and done by many other people. I learned that in order to make something new you have to be a critical thinker and put your own unique spin on something. Our group decided to make a common filter but we included the use of chloramines which is a less traditional purification method making our filter unique.
I will continue to work on spreading my work out evenly so I am not doing all of my work the day before an assignment is due. It is always best to work in smaller intervals instead of cramming in information so on the next project I will work on doing an even workload throughout the week. I can also improve on making sure we allow for more time to organize and practice our presentation. We can always improve on making sure our slide show presentation can flow smoother and this can be accomplished by setting aside one day to focus on practice presentations. A peak from this project was when our filter actually lowered the amount of iron present in the dirty water. All of our other tests did not give us any idea of whether or not our filter was doing anything but the iron levels did actually lower after being filtered. A pit from this project was when our filter broke. The paper that we used to line the bottom of the filter had deteriorated after many uses and much of the dirty water was able to pass through the filter. Even with this setback we were able to continue on and reconstruct a new filter to finish up the last few testes we needed to complete.