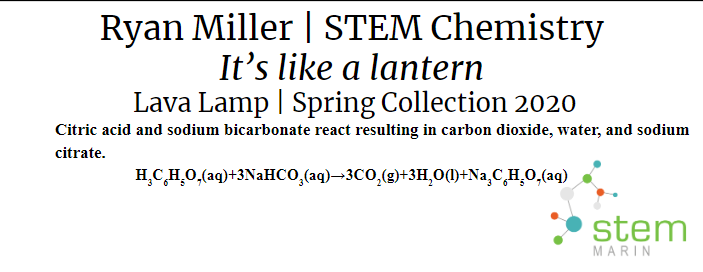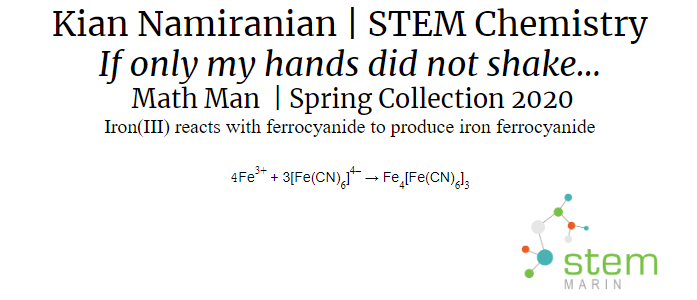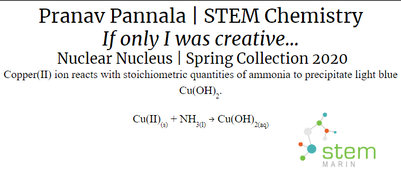Evidence of Work:
In this project we were tasked with creating a work of art using the chemistry that we had learned through the semester. Our group decided to focus on glow in the dark materials so that became the theme of our project. Next, we decided to each make an art piece focusing on the four letters in STEM: science, technology, engineering, and math. (Pictures can be found in the presentation below)
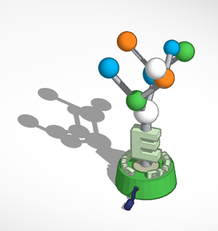
3D Sculpture:
|
To make the 3D Sculpture, I designed the model online using Tinkercad. I then 3D printed the parts required using different colored filament. I painted some of the parts and then glued them together to form the completed sculpture. In order to ensure the structural integrity of the model I ran wooden dowels through parts of the model and secured the pieces with super glue. The display card that will go with the sculpture is to the right. On the card the concept of luminescence is described along with the title and catch phrase for the art piece.
|
Lava Lamp:The lava lamp was created using oil and water within a clear container. The lamp can then be illuminated using a light and Alka Seltzer tablets can be added to create a chemical reaction which creates the bubbles you see in a lava lamp. The display card is shown below and the chemistry behind the reaction will be described in the content section.
|
Painting:The painting was created using Prussian blue paint that our group made. In order to make the paint we had to create a precipitate that we would mix with a paint base. The precipitate was created through a chemical reaction which will be explained in the content section. The painting was based on mathematician Georg Cantor and painted using glow in the dark paint so it would be seen at night.
|
Nuclear Nucleus:
Our Presentation:
Content:
Chemical Reaction:
A chemical reaction is a process when one or more substances are changed into one or more new substances. Chemical reactions were a key part of this project to produce the paint, oxidize the copper, and create the lava lamp bubbles.
A chemical reaction is a process when one or more substances are changed into one or more new substances. Chemical reactions were a key part of this project to produce the paint, oxidize the copper, and create the lava lamp bubbles.
|
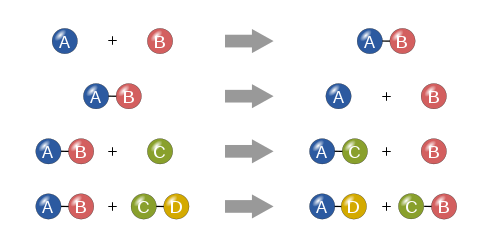
Types of Reactions:
There are five main types of chemical reactions. The first is a synthesis reaction. In a synthesis reaction two reactants produce one final product. An example of a synthesis reaction was when the copper was mixed with the ammonia. Copper(II) and ammonia reacted to create one final product: copper hydroxide. Another synthesis reaction was making the Prussian blue paint. Iron(III) reacted with ferrocyanide to produce iron ferrocyanide. The second type is a decomposition reaction. In a decomposition reaction one reactant is split into two products. In our project we did not use any decomposition reactions. The next type of reaction is a single replacement reaction. In a single replacement reaction one element replaces another element in one of the compounds in the reaction to produce new products. We did not use any single replacement reactions in our project. Double replacement reactions are another type of chemical reaction. In a double replacement reaction occurs when two ionic compounds exchange their ions to produce two new ionic compounds. In our project we used a special type of double replacement reaction called a neutralization reaction. In a neutralization reaction the reaction is between a base and an acid. This type of reaction occurred in the lava lamp where citric acid and sodium bicarbonate reacted to create carbon dioxide, water, and sodium citrate. The carbon dioxide that was created gave the lamp the bubbles to make it like a lava lamp. The final reaction type is a combustion reaction. Combustion reactions result in the burning of something and it produces water and carbon dioxide. Oxygen is also one of the reactants that goes into this type of reaction. We did not use any combustion reactions in our project. |
|
Balancing Chemical Reactions:
An important part of chemical reactions is knowing how to balance them. Because of the law of conservation of mass, chemical equations must always be balanced. You must have the same number of each element on each side of the chemical equation. For example, if you have four oxygen on the reactant side then you must have four oxygen on the product side as well. The charges in a chemical reaction must be balanced as well. For example, if you had aluminum oxide, you would require two aluminum and three oxygen. This is because aluminum had a +3 charge and oxide has a -2 charge. When you have the correct amount of each element the charges cancel and you have a balanced chemical reaction. |
|
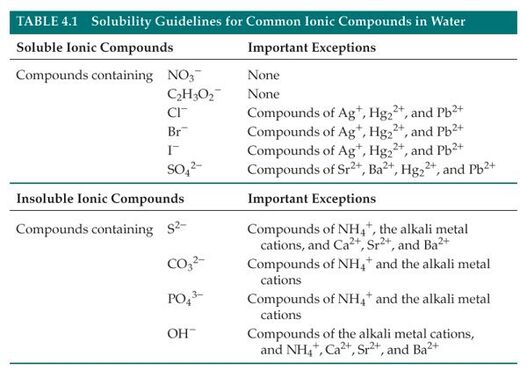
Precipitate:
When something is insoluble, it means that it cannot be dissolved into a liquid. Something that is soluble will dissolve into a liquid. In a precipitation reaction a precipitate is formed which is an example of something that is insoluble. When looking at a reaction, you will be able to tell if you have a soluble or insoluble ionic compound and whether or not a precipitate will form be looking at the solubility guidelines. When something is insoluble it means that everything did not dissolve. This means that a solid will have formed which is called a precipitate. When making paint, it was important that a precipitate formed when we were reacting iron with ferrocyanide. The precipitate that was formed form this reaction could then be used with a paint base to make Prussian blue paint. |
|
In order to produce oxidized copper, copper(II) reacted with ammonia to create a light blue precipitate (Cu(OH)2).
Chemical Reaction: Cu(II)(s) + NH3(l) → Cu(OH)2(aq) In order to produce the bubbles in the lava lamp, citric acid and sodium bicarbonate reacted resulting in carbon dioxide, water, and sodium citrate. Chemical Reaction: H3C6H5O7(aq)+3NaHCO3(aq) → 3CO2(g)+3H2O(l)+Na3C6H5O7(aq) In order to make the paint and 3D filament glow in the dark, strontium aluminate absorbed UV light. This then excited the electrons to higher energy orbitals. As the electrons slowly fell back to their lower orbital, photons were released which create a green glow. Reaction: UV Light + SrAl2O4 → Photons (520 nm) In order to make the blue paint, Iron(III) reacted with ferrocyanide to produce iron ferrocyanide. Chemical Reaction: 4Fe3+ 3[Fe(CN)6]4 → Fe4[Fe(CN)6]3 |
Prussian blue precipitate (above) and the copper blue (below)
|
Reflection:
One thing I did well on this project was my communication skills and my critical thinking. Throughout this project I did a lot of work outside of class. While working at home, I had to make sure that I was still working with the other members in my group. In order to ensure that the project would be successful, I had to communicate with my teammates to make sure that we had all of our work ready and presentable for the presentation. I also used my critical thinking skills when designing my sculpture. The weight of the 3D printed parts was too heavy for the base to support using only super glue. I had to find new solutions to fix the problem and I ended up creating wooden supports within the sculpture to ensure that it would not fall apart. A peak from this project was when we originally came up with our idea. We were all able to work together and collaborate in order to find an idea that interested all of us. A pit was towards the end of the project. We had a hard time transitioning from the building stage to the presentation stage which included making our slide show. The main part that went poorly was getting the slide show finished in the last few days. We had to do the slide show with less time than would have been ideal but in the end we were able to pull all of our work together.
I want to work on finishing parts of the project sooner or at home a least a few days before the presentation. This will allow my group in the future to practice or fine tune the presentation a few days before it is due. I also want to work on spacing my work load out evenly. In the future I will divide up what I have to do so it can be completed over a longer period of time. I learned that sometimes things are not as bad as they seem. I was originally skeptical of having to make an art piece but I found that there were many different options for this project. I was able to find an idea that interested me and did not require large amounts of artistic talent. Finally, I learned that perseverance is an important skill to have. Our paint did not work after multiple tries but we did not give up. We kept trying to make it work and we were eventually able to a paint that we could use on our art pieces.
I want to work on finishing parts of the project sooner or at home a least a few days before the presentation. This will allow my group in the future to practice or fine tune the presentation a few days before it is due. I also want to work on spacing my work load out evenly. In the future I will divide up what I have to do so it can be completed over a longer period of time. I learned that sometimes things are not as bad as they seem. I was originally skeptical of having to make an art piece but I found that there were many different options for this project. I was able to find an idea that interested me and did not require large amounts of artistic talent. Finally, I learned that perseverance is an important skill to have. Our paint did not work after multiple tries but we did not give up. We kept trying to make it work and we were eventually able to a paint that we could use on our art pieces.
Sources:
Research Sources:
https://www.amazon.com/NOVAMAKER-Filament-Dimensional-Accuracy-Luminous/dp/B0746FNJH6/ref=sr_1_9?keywords=glow+in+the+dark+pla&qid=1583269350&sr=8-9
https://www.stemjobs.com/chemistry-behind-glow-in-the-dark/
https://3dinsider.com/glow-in-the-dark-filament/
https://www.chemistryworld.com/podcasts/strontium-aluminate/3008606.article
Image Sources:
http://www.public.asu.edu/~jpbirk/qual/qualanal/copper.html
https://chemistryfanatics.wordpress.com/2013/07/02/experiment-8-prussian-blue-synthesis/
https://www.youtube.com/watch?v=lh8lzkXQO18
https://www.pinterest.co.uk/pin/141652350755277669/
https://en.wikipedia.org/wiki/Chemical_reaction
https://www.iconfinder.com/icons/4092499/chemical_chemical_reaction_experiment_flask_reaction_icon
https://www.amazon.com/NOVAMAKER-Filament-Dimensional-Accuracy-Luminous/dp/B0746FNJH6/ref=sr_1_9?keywords=glow+in+the+dark+pla&qid=1583269350&sr=8-9
https://www.stemjobs.com/chemistry-behind-glow-in-the-dark/
https://3dinsider.com/glow-in-the-dark-filament/
https://www.chemistryworld.com/podcasts/strontium-aluminate/3008606.article
Image Sources:
http://www.public.asu.edu/~jpbirk/qual/qualanal/copper.html
https://chemistryfanatics.wordpress.com/2013/07/02/experiment-8-prussian-blue-synthesis/
https://www.youtube.com/watch?v=lh8lzkXQO18
https://www.pinterest.co.uk/pin/141652350755277669/
https://en.wikipedia.org/wiki/Chemical_reaction
https://www.iconfinder.com/icons/4092499/chemical_chemical_reaction_experiment_flask_reaction_icon